
NBCn1 Increases NH4+ Reabsorption Across Thick Ascending Limbs, the Capacity for Urinary NH4+ Excretion, and Early Recovery from Metabolic Acidosis | American Society of Nephrology
![SOLVED: Draw the curved arrows for the mechanism of the following acid-base reaction. NH3 HSO3 5 NH4 SO32 - Marvin JS 0 ? ( * 0 0] @,+ + @ Help H c N 0 2 H S HsN: 1 A F a Br A SOLVED: Draw the curved arrows for the mechanism of the following acid-base reaction. NH3 HSO3 5 NH4 SO32 - Marvin JS 0 ? ( * 0 0] @,+ + @ Help H c N 0 2 H S HsN: 1 A F a Br A](https://cdn.numerade.com/ask_images/0735dfc0d6d84edcb687259d31624b4c.jpg)
SOLVED: Draw the curved arrows for the mechanism of the following acid-base reaction. NH3 HSO3 5 NH4 SO32 - Marvin JS 0 ? ( * 0 0] @,+ + @ Help H c N 0 2 H S HsN: 1 A F a Br A

⚗️HELP In the following acid-base reaction, NH4+ is the H2PO4- (aq) + NH3(aq) → HPO42- (aq) + - Brainly.com
Write a balanced equation for the dissociation of the following in water and identify the conjugate acid base pairs. - Sarthaks eConnect | Largest Online Education Community
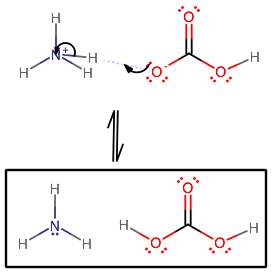
In the following acid-base reaction, how would you identify the acid, base, and their conjugate acids and bases: NH4+ + HCO3- --> NH3 + H2CO3? | Socratic

In the following acid-base reaction, NH4+ is the ______. 1. Base 2. Conjugate base 3. Conjugate - Brainly.com


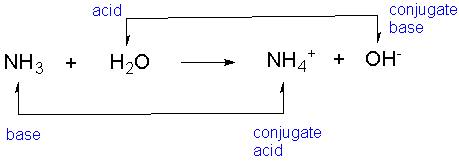

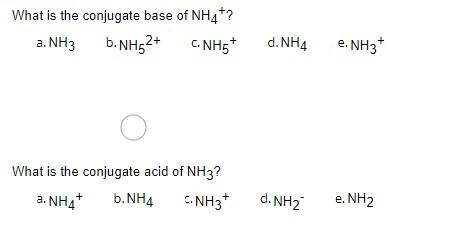




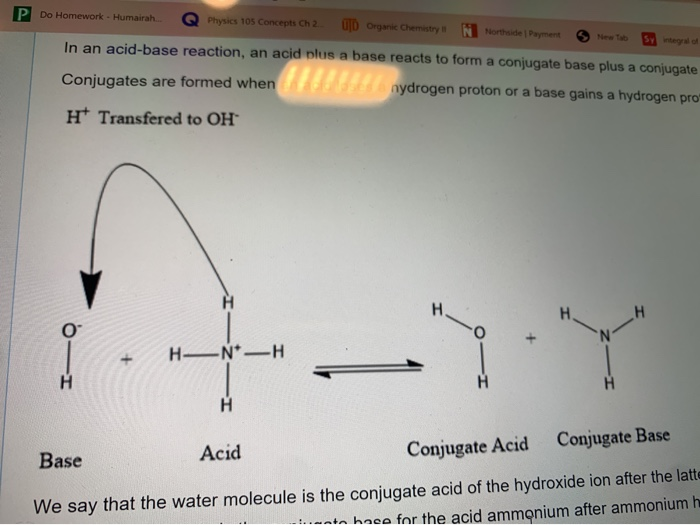
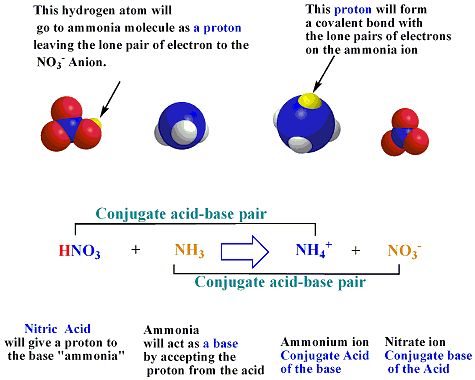


![PDF] Role of NH3 and NH4+ transporters in renal acid-base transport. | Semantic Scholar PDF] Role of NH3 and NH4+ transporters in renal acid-base transport. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/18fd1a5c866f58b0d9cc30a7046f9cba65cdddd7/3-Figure3-1.png)



