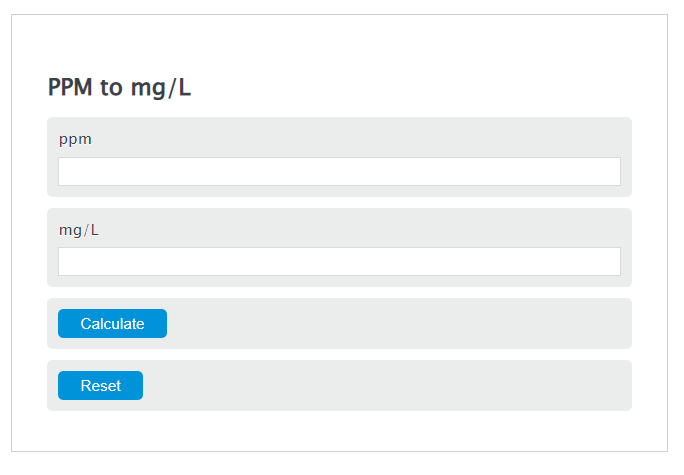
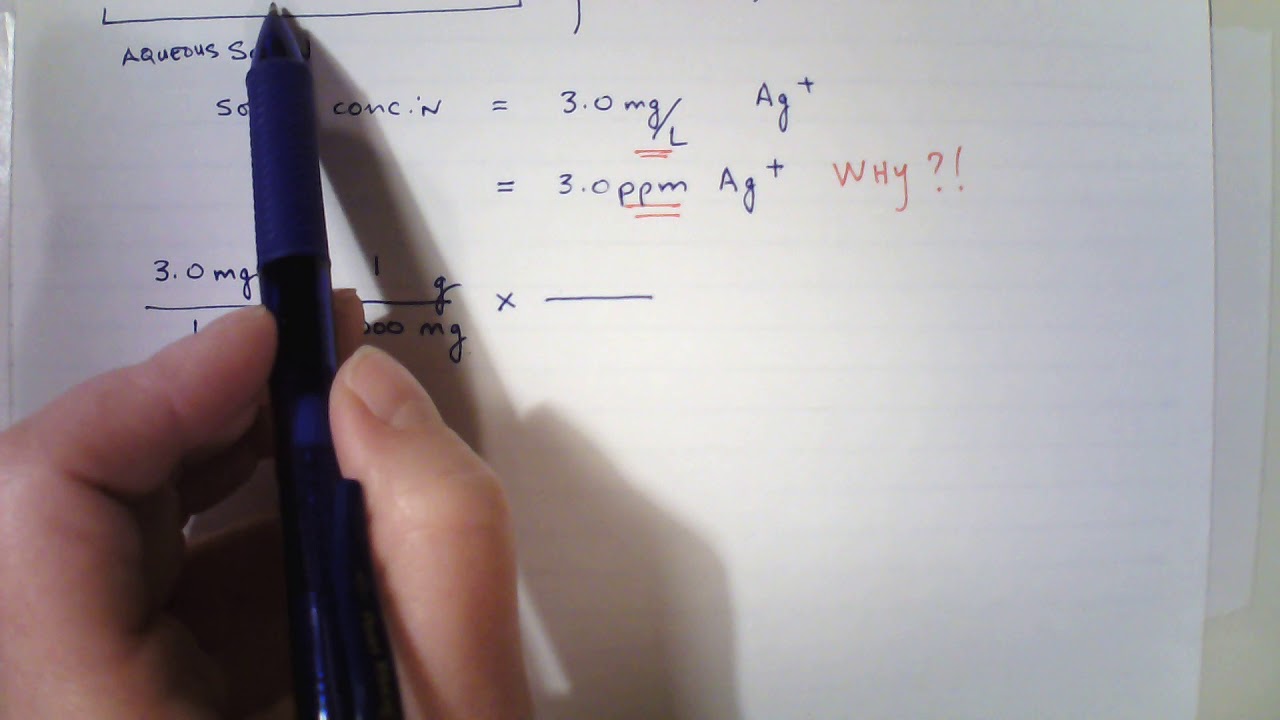How to convert arsenic concentration ug/L (in solution) to mg/kg (in soil) after sequential extraction experiment ? | ResearchGate

Pounds Formula Prepared By Michigan Department of Environmental Quality Operator Training and Certification Unit. - ppt download

SOLVED: Calculate the theoretical concentration of Fe2+ in ppm (mg/L) if 0.734 grams of Fe(NHa)(SO4)2 6HzO salt was weighed on an analytical balance and dissolved in a 1000.0 mL volumetric flask which