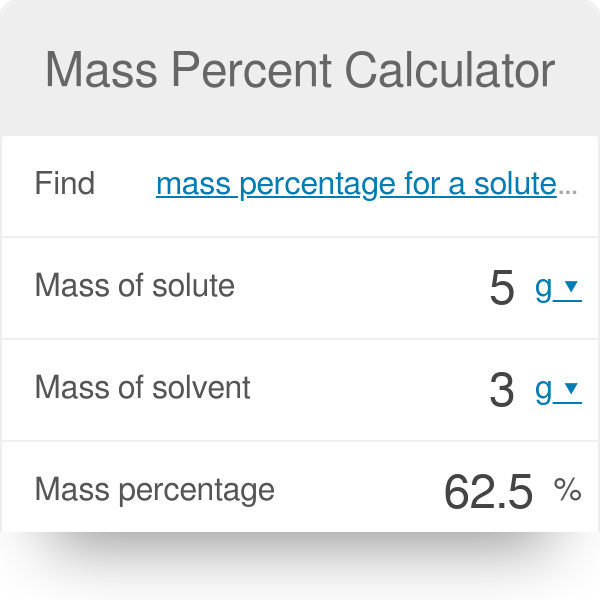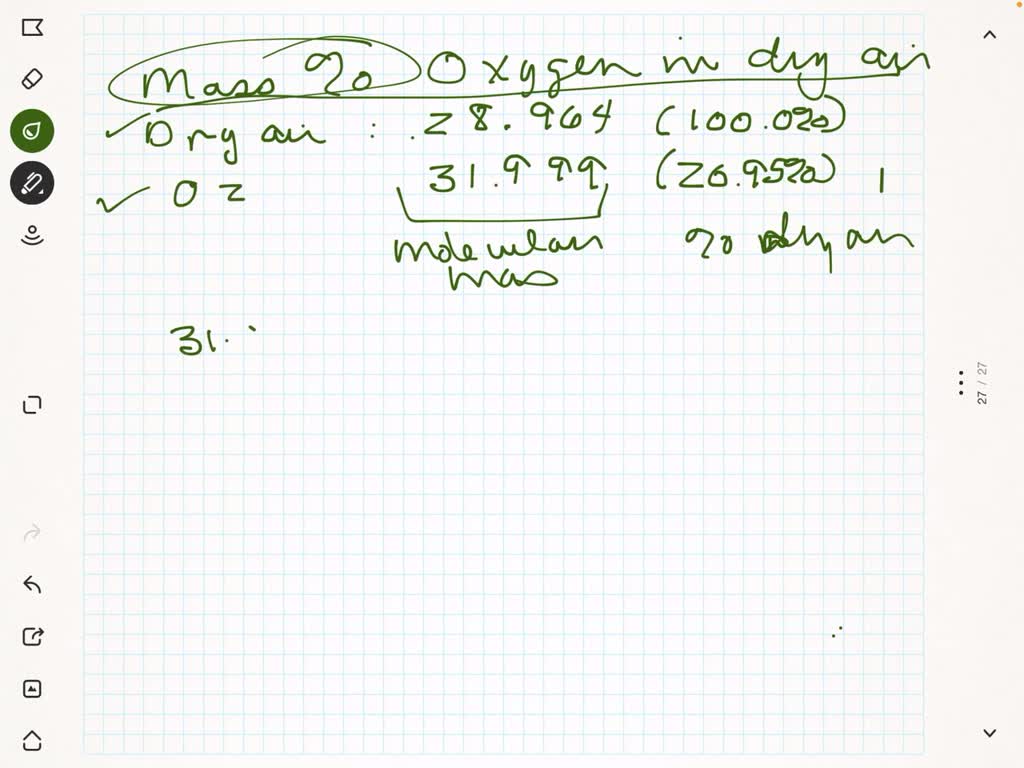
SOLVED: Part B Calculate the mass percentage of oxygen in dry air Express your answer with the appropriate units. View Available Hint(s) Value Units Submit

Calculate the mass percentage of carbon, hydrogen and oxygen in ethanol - CBSE Class 11 Chemistry - Learn CBSE Forum

SOLVED: Calculate mass percent composition of carbon in each compound. a.) C2H2 b.) C3H6 c.) C2H6 d.) C2H6O

Question Video: Calculating the Percent by Mass of the Active Ingredient in an Anti-inflammatory Ointment | Nagwa

SOLVED: Calculate the mass percent composition of carbon in each carbon-containing compound. a. CH4 b. C2H6 c. C2H2 d. C2H5Cl

How to Calculate Mass Percent of Element in Compound Examples, Practice Problem, Explained, Shortcut - YouTube


:max_bytes(150000):strip_icc()/Potassium-ferricyanide-58a279693df78c475811ba19.jpg)



:max_bytes(150000):strip_icc()/mass-percent-composition-example-609567_V2-01-89c18a9d30ea43b494d09b81f7ffefc1.png)