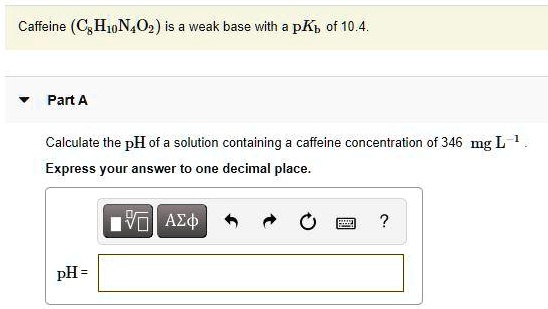
SOLVED: Caffeine (CgHjN,02) is weak base with pKb of 10.4 Part A Calculate the pH of a solution containing caffeine concentration of 346 mg L Express your answer to one decimal place.

0.070 g of caffeine is dissolved in 4.0 mL of water. The caffeine is extracted from the aqueous - YouTube